1.Explain the formation of a chemical bond.
Ans. According to Kossel and Lewis, atoms combine together in order to complete their respective octets so as to acquire the stable inert gas configuration. This can occur in two ways; by transfer of one or more electrons from one atom to other or by sharing of electrons between two or more atoms.
Ans. According to Kossel and Lewis, atoms combine together in order to complete their respective octets so as to acquire the stable inert gas configuration. This can occur in two ways; by transfer of one or more electrons from one atom to other or by sharing of electrons between two or more atoms.
2.Write Lewis dot symbols for atoms of the following elements: Mg, Na, B, O, N, Br.
Ans.

Ans.

3.Write Lewis symbols for the following atoms and ions: S and S2– ; Al and Al3+; H and H–
Ans.
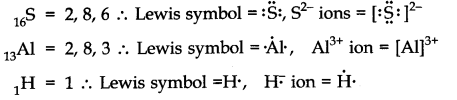
Ans.
4.Draw the Lewis structures for the following molecules and ions:
H2S, SiCl4 , BeF2, C032-, HCOOH
Ans.
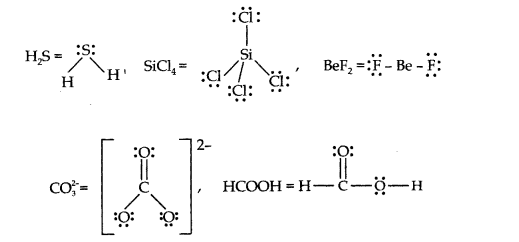
H2S, SiCl4 , BeF2, C032-, HCOOH
Ans.

5.Define Octet rule. Write its significance and limitations.
Ans. Octet rule: Atoms of elements combine with each other in order to complete their respective octets so as to acquire the stable gas configuration.
Significance: It helps to explain why different atoms combine with each other to form ionic compounds or covalent compounds.
Limitations of Octet rule:
(i)According to Octet rule, atoms take part in chemical combination to achieve the configuration of nearest noble gas elements. However, some of noble gas elements like Xenon have formed compounds with fluorine and oxygen. For example:XeF2, XeF4etc.
Therefore, validity of the octet rule has been challenged.
(ii)This theory does not account for shape of molecules.
Ans. Octet rule: Atoms of elements combine with each other in order to complete their respective octets so as to acquire the stable gas configuration.
Significance: It helps to explain why different atoms combine with each other to form ionic compounds or covalent compounds.
Limitations of Octet rule:
(i)According to Octet rule, atoms take part in chemical combination to achieve the configuration of nearest noble gas elements. However, some of noble gas elements like Xenon have formed compounds with fluorine and oxygen. For example:XeF2, XeF4etc.
Therefore, validity of the octet rule has been challenged.
(ii)This theory does not account for shape of molecules.
6. Write the favourable factors for the formation of ionic bond.
Ans. (i) Low ionization enthalpy of metal atoms
(ii) High electron gain enthalpy of non-metal atoms (iii) High lattice enthalpy of compound formed.
Ans. (i) Low ionization enthalpy of metal atoms
(ii) High electron gain enthalpy of non-metal atoms (iii) High lattice enthalpy of compound formed.
7. Discuss the shape of the following molecules using the VSEPR model:
BeCl2, BCl3 , SiCl4, AsF5, H2S, PH3
Ans.

BeCl2, BCl3 , SiCl4, AsF5, H2S, PH3
Ans.

8.Although geometries of NH3 and H20 molecules are distorted tetrahedral, bond angle in water is less than that of ammonia. Discuss.
Ans.
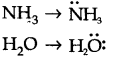
Because of two lone pairs of electrons on O-atom, repulsion on bond pairs is greater in H20 in comparison to NH3 . Thus, the bond angle is less in H20 molecules.
Ans.
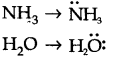
Because of two lone pairs of electrons on O-atom, repulsion on bond pairs is greater in H20 in comparison to NH3 . Thus, the bond angle is less in H20 molecules.
9.How do you express the bond strength in terms of bond order?
Ans. Bond strength is directly proportional to the bond order. Greater the bond order, more is the bond strength.
Ans. Bond strength is directly proportional to the bond order. Greater the bond order, more is the bond strength.
10. Define the bond-length.
Ans. Bond-length: It is the equilibrium distance between the nuclei of two bonded atoms in a molecule. Bond-lengths are measured by spectroscopic methods.
Ans. Bond-length: It is the equilibrium distance between the nuclei of two bonded atoms in a molecule. Bond-lengths are measured by spectroscopic methods.
11.Explain the important aspects of resonance with reference to the C032-ion.
Ans.
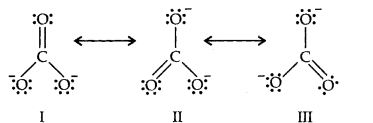
Resonance in C032-, I, II and III represent the three canonical forms.
(i)In these structures, the position of nuclei are same.
(ii)All the three forms have almost equal energy.
(iii)Same number of paired and impaired electrons, they differ only in their position.
Ans.

Resonance in C032-, I, II and III represent the three canonical forms.
(i)In these structures, the position of nuclei are same.
(ii)All the three forms have almost equal energy.
(iii)Same number of paired and impaired electrons, they differ only in their position.
12. H3 P03can be represented by structures 1 and 2 shown below. Can these two structures be taken as the canonical forms of the resonance hybrid representing H3 P03? If not, give reasons for the same.
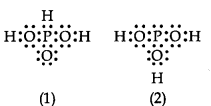
Ans.No, these cannot be taken as canonical forms because the positions of atoms have been changed.
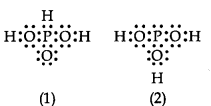
Ans.No, these cannot be taken as canonical forms because the positions of atoms have been changed.
13.Write the resonance structures for SO3,N02 and N03
Ans.
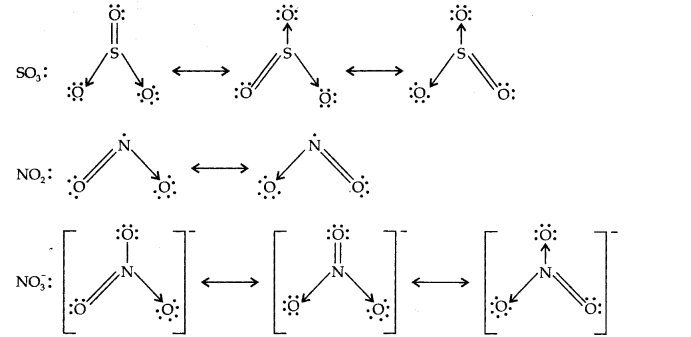
Ans.

14.Use Lewis symbols to show electron transfer between the following atoms to form cations and anions (a) K and S (b) Ca and O (c) Al and N.
Ans.
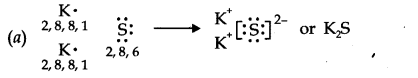
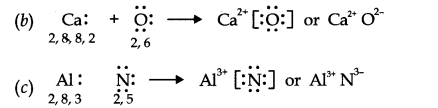
Ans.
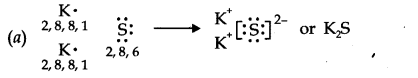
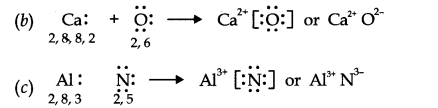
15.Although both C02 and H20 are triatomic molecules, the shape of H20 molecule is bent while that of C02 is linear. Explain this on the basis of dipole moment.
Ans. In C02, there are two C=0 bonds. Each C=0 bond is a polar bond.
The net dipole moment of C02 molecule is zero. This is possible only if C02 is a linear molecule. (0=C=0). The bond dipoles of two C=0 bonds cancel the moment of each other.
Whereas, H20 molecule has a net dipole moment (1.84 D). H20 molecule has a bent structure because here the O—H bonds are oriented at an angle of 104.5° and do not cancel the bond moments of each other.
Ans. In C02, there are two C=0 bonds. Each C=0 bond is a polar bond.
The net dipole moment of C02 molecule is zero. This is possible only if C02 is a linear molecule. (0=C=0). The bond dipoles of two C=0 bonds cancel the moment of each other.
Whereas, H20 molecule has a net dipole moment (1.84 D). H20 molecule has a bent structure because here the O—H bonds are oriented at an angle of 104.5° and do not cancel the bond moments of each other.
16.Write the significance/applications of dipole moment.
Ans. (i)In predicting the nature of the molecules: Molecules with specific dipole moments are polar in nature and those of zero dipole moments are non-polar in nature.
(ii)In the determination of shapes of molecules.
(iii)In calculating the percentage ionic character.
Ans. (i)In predicting the nature of the molecules: Molecules with specific dipole moments are polar in nature and those of zero dipole moments are non-polar in nature.
(ii)In the determination of shapes of molecules.
(iii)In calculating the percentage ionic character.
17.Define electronegativity. How does it differ from electron gain enthalpy?
Ans. Electronegativity: Electronegativity is the tendency of an atom to attract shared pair of electrons. It is the property of bonded atom.
Whereas, electron gain enthalpy is the tendency of an atom to attract outside electron. It is the property of an isolated atom.
Ans. Electronegativity: Electronegativity is the tendency of an atom to attract shared pair of electrons. It is the property of bonded atom.
Whereas, electron gain enthalpy is the tendency of an atom to attract outside electron. It is the property of an isolated atom.
18.Explain with the help of suitable example polar covalent bond.
Ans. When two atoms with different electronegativity are linked to each other by covalent bond, the shared electron pair will not in the centre because of the difference in electronegativity. For example, in hydrogen flouride molecule, flouride has greater electronegativity than hydrogen. Thus, the shared electron pair is displaced more towards’flourine atom, the later will acquire a partial negative charge (∂–). At the same time hydrogen atom will have a partial positive charge (∂+). Such a covalent bond is known as polar covalent bond or simply polar bond.
It is represented as
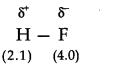
Ans. When two atoms with different electronegativity are linked to each other by covalent bond, the shared electron pair will not in the centre because of the difference in electronegativity. For example, in hydrogen flouride molecule, flouride has greater electronegativity than hydrogen. Thus, the shared electron pair is displaced more towards’flourine atom, the later will acquire a partial negative charge (∂–). At the same time hydrogen atom will have a partial positive charge (∂+). Such a covalent bond is known as polar covalent bond or simply polar bond.
It is represented as
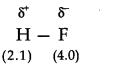
19. Arrange the bonds in order of increasing ionic character in the molecules:LiF, K20, N2, S02 and CIF3.
Ans.N2 < S02 < ClF3 < K20 < LiF
Ans.N2 < S02 < ClF3 < K20 < LiF
20.The skeletal structure of CH3COOH as shown below is correct, but some of the bonds are shown incorrectly. Write the correct Lewis structure for acetic acid.
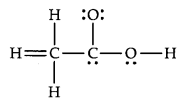
Ans.
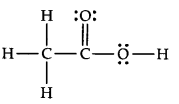
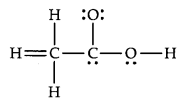
Ans.
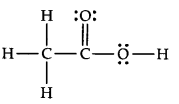
21.Apart from tetrahedral geometry, another possible geometry for CH4 is square planar with the four H atoms at the comers of the square and the C atom at its centre. Explain why CH4 is not square planar?
Ans.According to VSEPR theory, if CH4were square planar, the bond angle would be 90°. For tetrahedral structure, the bond angle is 109°28′. Therefore, in square planar structure, repulsion between bond pairs would be more and thus the stability will be less.
Ans.According to VSEPR theory, if CH4were square planar, the bond angle would be 90°. For tetrahedral structure, the bond angle is 109°28′. Therefore, in square planar structure, repulsion between bond pairs would be more and thus the stability will be less.
22.Explain why BeH2 molecule has a zero dipole moment although the Be—H bonds are polar.
Ans. BeH2is a linear molecular (H—Be—H), the bond angle = 180°.
Be—H bonds are polar due to difference in their electronegativity but the bond polarities cancel each other. Thus, molecule has resultant dipole moment of zero.
Ans. BeH2is a linear molecular (H—Be—H), the bond angle = 180°.
Be—H bonds are polar due to difference in their electronegativity but the bond polarities cancel each other. Thus, molecule has resultant dipole moment of zero.
23.Which out of NH3 and NF3 has higher dipole moment and why?
Ans.In NH3 and NF3, the difference in electronegativity is nearly same but the dipole moment of NH3 = (1.46D) For Example, NH3 = (0.24D)
In NH3, the dipole moments of the three N—H bonds are in the same direction as the lone pair of electron. But in NF3, the dipole moments of the three N—F bonds are in the direction opposite to that of the lone pair. Therefore, the resultant dipole moment in NH3 is more than in NF3.
Ans.In NH3 and NF3, the difference in electronegativity is nearly same but the dipole moment of NH3 = (1.46D) For Example, NH3 = (0.24D)
In NH3, the dipole moments of the three N—H bonds are in the same direction as the lone pair of electron. But in NF3, the dipole moments of the three N—F bonds are in the direction opposite to that of the lone pair. Therefore, the resultant dipole moment in NH3 is more than in NF3.
24.What is meant by hybridisation of atomic orbitals? Describe the shapes of sp, SP2, SP3 hybrid orbitals.
Ans. Hybridisation: It is defined as the process of intermixing of atomic oribitals of slightly different energies to give rise to new hybridized orbitals having equivalent energy and identical shapes.
Shapes of Orbitals:
sp hybridisation: When one s-and one p-orbital, intermix then it is called sp-hybridisation. For example, in BeF2, Be atom undergoes sp-hybridisation. It has linear shape. Bond angle is 180°.
SP2hybridisation: One s-and two p-orbitals get hybridised to form three equivalent hybrid orbitals. The three hybrid orbitals directed towards three corners of an equilateral triangle. It is, therefore, known as trigonal hybridisation.
SP3 hybridisation: One s-and three p-orbitals get hybridised to form four equivalent hybrid orbitals. These orbitals are directed towards the four corners of a regular tetrahedron.
Ans. Hybridisation: It is defined as the process of intermixing of atomic oribitals of slightly different energies to give rise to new hybridized orbitals having equivalent energy and identical shapes.
Shapes of Orbitals:
sp hybridisation: When one s-and one p-orbital, intermix then it is called sp-hybridisation. For example, in BeF2, Be atom undergoes sp-hybridisation. It has linear shape. Bond angle is 180°.
SP2hybridisation: One s-and two p-orbitals get hybridised to form three equivalent hybrid orbitals. The three hybrid orbitals directed towards three corners of an equilateral triangle. It is, therefore, known as trigonal hybridisation.
SP3 hybridisation: One s-and three p-orbitals get hybridised to form four equivalent hybrid orbitals. These orbitals are directed towards the four corners of a regular tetrahedron.
25.Describe the change in hybridisation (if any) of the Al atom in the following reaction.
AlCl3 + Cl– ——>AlCl4- .
Ans. Electronic configuration of 13Al = 1s2 2s2 2p6 3s1 3px13py1
(excited state)
Hence, hybridisation will be SP2
In AlCl–4, the empty 3pzorbital is also involved. So, the hybridisation is SP3 and the shape is tetrahedral.
AlCl3 + Cl– ——>AlCl4- .
Ans. Electronic configuration of 13Al = 1s2 2s2 2p6 3s1 3px13py1
(excited state)
Hence, hybridisation will be SP2
In AlCl–4, the empty 3pzorbital is also involved. So, the hybridisation is SP3 and the shape is tetrahedral.
26. Is there any change in the hybridisation ofB and N atoms as a result of the following reaction ?BF3 + NH3 ——-> F3 B.NH3
Ans. In BF3, B atom is SP2 hybridised. In NH3, N is SP3 hybridised.
After the reaction, hybridisation of B changes from SP2 to SP3.
Ans. In BF3, B atom is SP2 hybridised. In NH3, N is SP3 hybridised.
After the reaction, hybridisation of B changes from SP2 to SP3.
27. Draw diagrams showing the formation of a double bond and a triple bond between carbon atoms in C2H4 and C2 H2 molecules.
Ans.
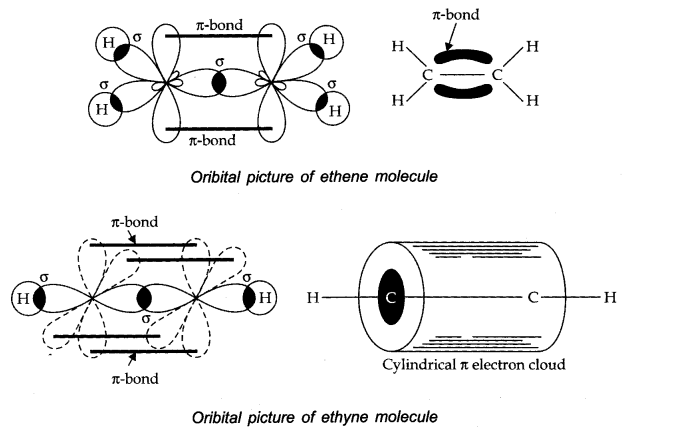
Ans.

28.What is the total number of sigma and pi bonds in the following molecules?
(a) C2 H2 (b)C2 H4
Ans. (a) H—C = C—H
Sigma bond = 3 Π bonds = 2
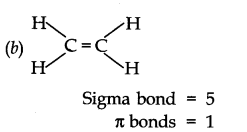
(a) C2 H2 (b)C2 H4
Ans. (a) H—C = C—H
Sigma bond = 3 Π bonds = 2

29.Considering X-axis as the intemuclear axis which out of the following will not form a sigma bond and why? (a) Is and Is (b) Is and 2px (c)2py and 2py (d) Is and 2s
Ans. (c) It will not form a s-bond because taking x-axis as the intemuclear axis, there will be lateral overlap between the two 2py orbitals forming a Π -bond.
Ans. (c) It will not form a s-bond because taking x-axis as the intemuclear axis, there will be lateral overlap between the two 2py orbitals forming a Π -bond.
30. Which hybrid orbitals are used by carbon atoms in the following molecules?
(a) CH3-CH3 (b) CH3-CH = CH2 (c) CH3-CH2-OH (d) CH3-CHO (e) CH3COOH.
Ans.
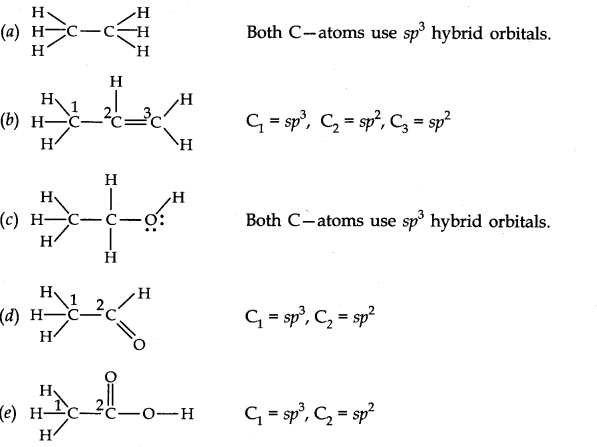
(a) CH3-CH3 (b) CH3-CH = CH2 (c) CH3-CH2-OH (d) CH3-CHO (e) CH3COOH.
Ans.

31.What do you understand by bond pairs and lone pairs of electrons? Illustrate by giving one example of each type.
Ans. The electron pair involved in sharing between two atoms during covalent bonding is called shared pair or bond pair. At the same time, the electron pair which is not involved in sharing is called lone pair of electrons.

Ans. The electron pair involved in sharing between two atoms during covalent bonding is called shared pair or bond pair. At the same time, the electron pair which is not involved in sharing is called lone pair of electrons.

32.Distinguish between a sigma bond and a pi bond.
Ans.

Ans.

33.Explain the formation of H2 molecule on the basis of valence bond theory.
Ans. Let us consider the combination between atoms of hydrogen HA and HB and eA and eB be their respective electrons.
As they tend to come closer, two different forces operate between the nucleus and the electron of the other and vice versa. The nuclei of the atoms as well as their electrons repel each other. Energy is needed to overcome the force of repulsion. Although the number of new attractive and repulsive forces is the same, but the magnitude of the attractive forces is more. Thus, when two hydrogen atoms approach each other, the overall potential energy of the system decreases. Thus, a stable molecule of hydrogen is formed.
Ans. Let us consider the combination between atoms of hydrogen HA and HB and eA and eB be their respective electrons.
As they tend to come closer, two different forces operate between the nucleus and the electron of the other and vice versa. The nuclei of the atoms as well as their electrons repel each other. Energy is needed to overcome the force of repulsion. Although the number of new attractive and repulsive forces is the same, but the magnitude of the attractive forces is more. Thus, when two hydrogen atoms approach each other, the overall potential energy of the system decreases. Thus, a stable molecule of hydrogen is formed.
34.Write the important conditions required for the linear combination of atomic orbitals to form molecular orbitals.
Ans. (i) The combining atomic orbitals should have comparable energies.
For example, Is orbital of one atom can combine with Is atomic orbital of another atom, 2s can combine with 2s.
(ii) The combining atomic orbitals must have proper orientations. So that they are able to overlap to a considerable extent.
(iii) The extent of overlapping should be large.
Ans. (i) The combining atomic orbitals should have comparable energies.
For example, Is orbital of one atom can combine with Is atomic orbital of another atom, 2s can combine with 2s.
(ii) The combining atomic orbitals must have proper orientations. So that they are able to overlap to a considerable extent.
(iii) The extent of overlapping should be large.
35.Use molecular orbital theory to explain why the Be2 molecule does not exist.
Ans.
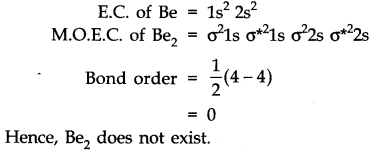
Ans.
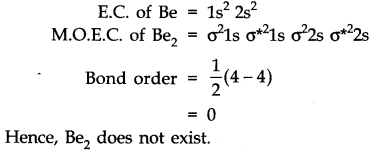
36.Compare the relative stability of the following species and indicate their magnetic properties: O2, O2, O2– (Superoxide),O22- (peroxide)
Ans.O2— Bond order = 2, paramagnetic
O2+— Bond order = 2.5, paramagnetic
O2–— Bond order = 1.5, paramagnetic
O22- — Bond order = 1, diamagnetic
Order of relative stability is
O2+ > O2>O2– > O22-
(2.5) (2.0) (1.5) (1.0)
Ans.O2— Bond order = 2, paramagnetic
O2+— Bond order = 2.5, paramagnetic
O2–— Bond order = 1.5, paramagnetic
O22- — Bond order = 1, diamagnetic
Order of relative stability is
O2+ > O2>O2– > O22-
(2.5) (2.0) (1.5) (1.0)
37.Write the significance of plus and minus sign in representing the orbitals,
Ans. Plus and minus sign is used to indentify the nature of electrons wave. Plus (+ve) sign denotes crest, while (-ve) sign denotes trough.
Ans. Plus and minus sign is used to indentify the nature of electrons wave. Plus (+ve) sign denotes crest, while (-ve) sign denotes trough.
38.Describe the hybridisation in case of PCl5. Why are the axial bonds longer as compared to equatorial bonds?
Ans. The ground state E.C. and the excited state E.C. of phosphorus are represented as:
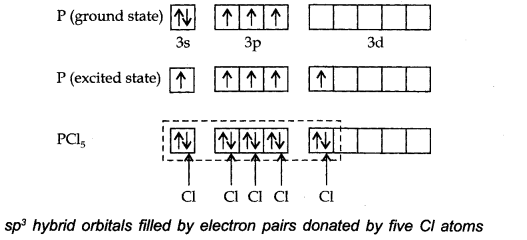
The one s, three-p and one d-orbitals hybridise to yield five sets of SP3 d hybrid orbitals which are directed towards the five corners of a trigonal bipyramidal as in Fig.
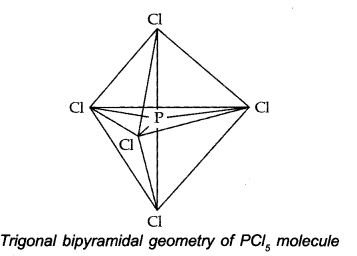
Because axial bond pairs suffer more repulsive interaction from the equatorial bond pairs, therefore axial bonds have been found to be slightly longer and hence slightly weaker than equatorial bonds.
Ans. The ground state E.C. and the excited state E.C. of phosphorus are represented as:

The one s, three-p and one d-orbitals hybridise to yield five sets of SP3 d hybrid orbitals which are directed towards the five corners of a trigonal bipyramidal as in Fig.

Because axial bond pairs suffer more repulsive interaction from the equatorial bond pairs, therefore axial bonds have been found to be slightly longer and hence slightly weaker than equatorial bonds.
39.Define hydrogen bonds. Is it weaker or stronger than the van der Waals forces?
Ans. When hydrogen is attached with highly electronegative element in a covalent bonding the electrons of the covalent bond are shifted towards the more electronegative atom. Thus, partially positively charged hydrogen atom forms a bond with the other more electronegative atom. This bond is known as hydrogen bond. Hydrogen bond is stronger than the van der Waals forces.
Ans. When hydrogen is attached with highly electronegative element in a covalent bonding the electrons of the covalent bond are shifted towards the more electronegative atom. Thus, partially positively charged hydrogen atom forms a bond with the other more electronegative atom. This bond is known as hydrogen bond. Hydrogen bond is stronger than the van der Waals forces.
40.What is meant by the term bond order? Calculate the bond order of:N2, O2, O2+,O2–
Ans. Bond order is defined as the half of the difference between the number of electrons present in bonding and antibonding molecular orbitals.

Ans. Bond order is defined as the half of the difference between the number of electrons present in bonding and antibonding molecular orbitals.



